The technology behind the Talis Biomedical One molecular point-of-care test
A new paradigm for infectious diseases molecular testing
At Talis Biomedical, we are pioneering a new paradigm for infectious diseases testing, aiming to bring the accuracy of a central lab molecular test to a variety of health care settings. The Talis Biomedical One Test System† is designed to allow healthcare providers to confidently run point-of-care-tests. Our system is designed with the goal of delivering accurate, sensitive results quickly due to the unique engineering and design of our nucleic acid amplification test (NAAT) chemistry.

HOW TALIS ONE MOLECULAR POINT-OF-CARE TEST TECHNOLOGY WORKS LEVERAGING LAMP FOR SPEED AND ACCURACY
The Talis Biomedical One System relies on real-time loop-mediated isothermal amplification (rtLAMP) for DNA targets and real-time reverse transcription loop-mediated isothermal amplification (rtRT-LAMP) for RNA targets (Figure 1).
Loop-mediated isothermal Amplification (LAMP) is a well-studied technique known for sensitivity and specificity, especially when nucleic acid is first extracted from the sample.1 LAMP is also fairly resistant to inhibitors in the sample,1 making it a good technique for diagnostic applications.
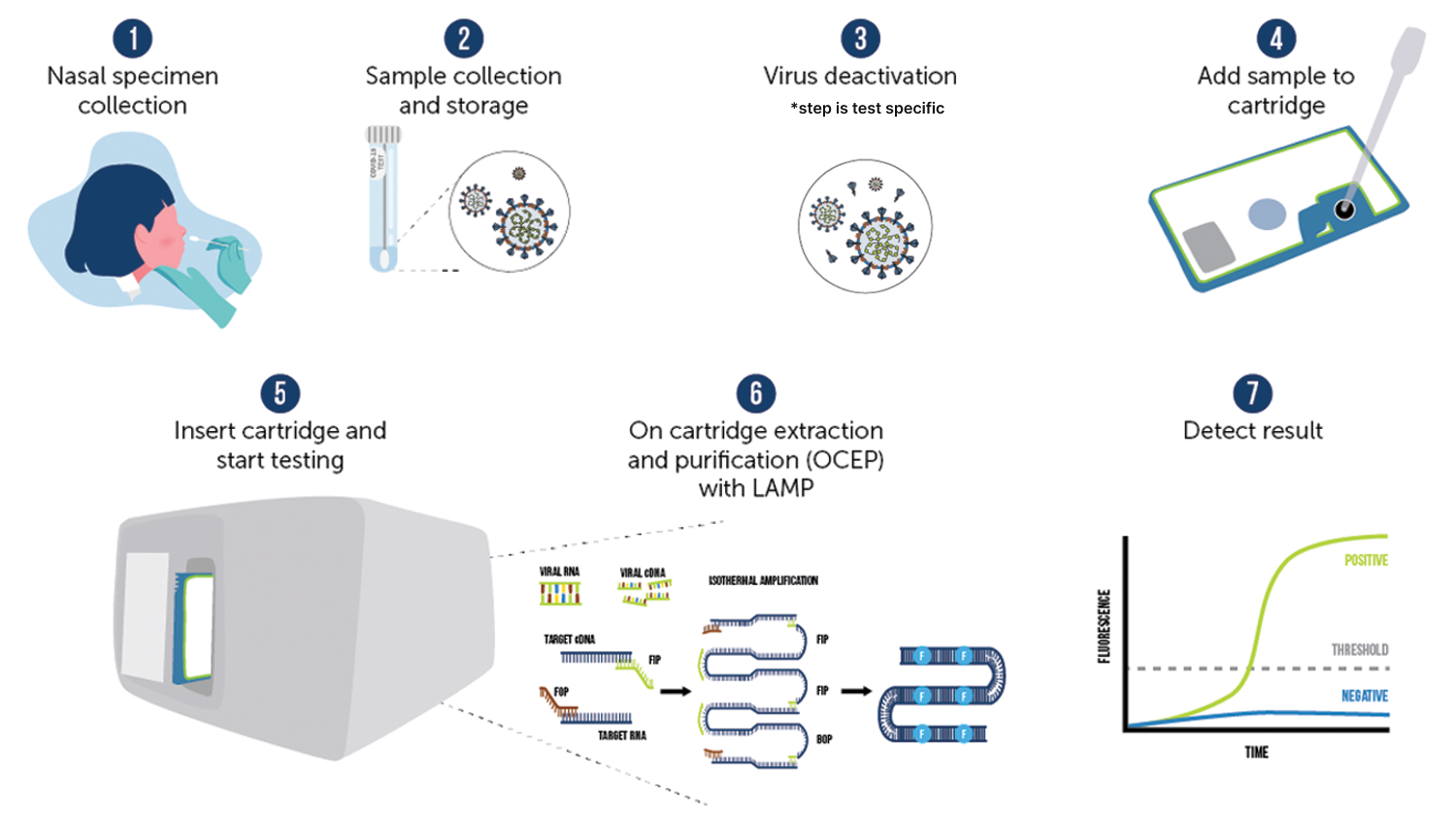
Figure 1.
WE’RE BRINGING MOLECULAR TESTING TO THE POINT OF CARE
While there are many kinds of nucleic acid amplification technologies (NAAT), many are not suited to point-of-care applications. Conventional polymerase chain reaction (PCR) based methods like qPCR need to cycle between multiple temperatures 20 or more times, which results in turnaround times of approximately 4 hours.1 Isothermal NAAT uses biochemistry that operates at a single temperature; therefore, these tests can deliver accurate results on a much faster timeline.
We are Optimizing Lamp Sensitivity
By including an on-cartridge sample extraction step, we’ve improved the sensitivity of LAMP. Our proprietary OCEP™ technology uses a unique sample extraction method via a solid matrix column-based approach which maximizes nucleic acid recovery, and the fully enclosed design of the Talis Biomedical One cartridge ensures that all fluids remain contained within the cartridge.
TALIS TECHNOLOGY IS EASY TO USE
Talis Biomedical One’s easy-to-use, sample-to-answer workflow enables infectious disease testing by healthcare professionals in a variety of point-of-care settings. The single-use test cartridge is a critical part of the system and where all the LAMP reactions take place. One of the key advantages of our testing workflow is the inclusion of a solid phase sample extraction step, to maximize the sensitivity of our system.
© Talis Biomedical Corporation. All Rights Reserved.
Talis One is a registered trademark of Talis Biomedical Corporation.
* Testing solutions are currently in development and not available for sale.
* †The Talis Biomedical One Test System is not authorized, cleared, or approved by the FDA and is not available for sale.
References
- Khan P, Aufdembrink LM, Engelhart AE. Isothermal SARS-CoV-2 Diagnostics: Tools for Enabling Distributed Pandemic Testing as a Means of Supporting Safe Reopenings. ACS Synth Biol. Published online September 23, 2020:acssynbio.0c00359. doi:10.1021/acssynbio.0c00359